Thank you. Listen to this article using the player above. ✖
The distribution of outermost shell electrons, known as valence electrons, of organic molecules was experimentally observed for the first time by a team led by Nagoya University in Japan. As the interactions between atoms are governed by the valence electrons, their findings shine light on the fundamental nature of chemical bonds, with implications for pharmacy and chemical engineering. The results were published in the Journal of the American Chemical Society.
The behavior of the electrons in atoms is complex, forming electron orbitals that have different functions depending on their closeness to the nucleus. The inner shell electrons, called core electrons, are used for self-stabilization and do not interact with other atoms. On the other hand, the outer electrons, or valence electrons, define most of the material’s properties, especially during bonding with other atoms.
Want more breaking news?
Subscribe to Technology Networks’ daily newsletter, delivering breaking science news straight to your inbox every day.
Understanding a material’s properties requires extracting information about its valence electrons. However, it has been difficult to experimentally isolate only the valence electron information, leading to researchers having to rely on theoretical models and spectroscopy to estimate it.
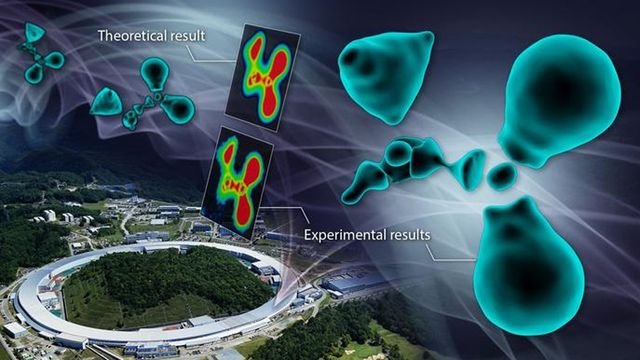
By conducting world-class synchrotron X-ray diffraction experiments at SPring-8, the group discovered that it is possible to selectively extract only the valence electron density of atoms in a crystal.
“We named this method the CDFS method. Using this method, we observed the electron state of the glycine molecule, a type of amino acid,” corresponding author Hiroshi Sawa said. “Although the method was relatively simple to perform, the result was impressive. The observed electron cloud did not exhibit the smooth, enveloping shape that many predicted, but rather a fragmented, discrete state.”
To understand the nature of the results, the group made a color map of their observations. In chemistry, a color map uses colors to display variations in datasets across a specific range. Such maps are often used in conjunction with spectroscopic techniques, imaging, and chemical analysis to provide an intuitive way to interpret complex datasets.
The map of the cross-sectional view in the enlarged diagram clearly showed interruptions in the electron distribution surrounding the carbon atoms.“When carbon forms bonds with surrounding atoms, it reconstructs its electron cloud to create hybridized orbitals. In this case, the outermost L-shell electrons have nodes based on their wave nature known as wave functions,” Sawa explained. “This means that due to the wave nature of electrons, there are parts of the hybrid orbitals where electrons are absent, much different to the image that many people have of a continuous ‘cloud’ of electrons.”
The fragmented electron cloud distribution observed in the experiment demonstrates the quantum mechanical wave nature of electrons, as predicted by physics. To confirm whether the observed electron cloud accurately captures the true state, they conducted advanced quantum chemical calculations in collaboration with Hokkaido University that confirmed that the experimental and theoretical results matched perfectly.
Sawa believes that the results show the benefits of interdisciplinary research. “I believe it has been helpful in providing a clear conclusion to the ambiguous understanding of bonding states that has puzzled researchers since the 19th century,” Sawa said. “Visualizing electron behavior is a challenging endeavor, yet the results can be elegantly understood as electrons acting in accordance with wave functions. I believe our findings have astonished many researchers and validated the model proposed by quantum chemistry.”
With a precise understanding of the valence electron density distribution forming this molecule, the group conducted similar experiments and calculations on cytidine, a slightly more complex molecule. They successfully extracted the electrons within the carbon double bonds and clearly observed differences between the carbon-carbon and carbon-nitrogen bonds.
“This study has made it possible to directly visualize the essence of chemical bonds, potentially contributing to the design of functional materials and the understanding of reaction mechanisms. This is because it aids in discussing the electronic states of molecules, which are difficult to infer from just the chemical structural formula,” Sawa said. “It may, for example, explain why some medicines work and others don’t. Fields where interactions influence functionality and structural stability, such as organic semiconductors and research on the structure of DNA double helixes, are likely to benefit most from our research.”
Reference: Hara T, Hasebe M, Tsuneda T, et al. Unveiling the nature of chemical bonds in real space. J Am Chem Soc. 2024:jacs.4c05673. doi: 10.1021/jacs.4c05673
This article has been republished from the following materials. Note: material may have been edited for length and content. For further information, please contact the cited source. Our press release publishing policy can be accessed here.